The animal production industry is in the middle of a fundamental shift. Reducing and replacing antibiotics is no longer aspirational — it is a commercial and regulatory reality. The challenge for producers is finding alternatives that actually deliver, without compromising performance or creating new problems down the line.
ALMAT, from our partners at Animal Nutrition Development (AND), represents one of the most advanced responses to that challenge. It works at a molecular level, targeting harmful bacteria with precision while leaving the beneficial gut microbiota intact — and the science behind it is worth understanding.

The Problem with Traditional Antibiotics
Conventional antibiotics work by eliminating susceptible bacteria broadly — which means they take out beneficial gut flora alongside the pathogens. The bacteria that survive are the resistant ones. Over time, this creates a cycle of microbial imbalance, reduced gut resilience, and diminishing returns from repeated treatments.
The industry has long needed tools that are more targeted. Antimicrobial Peptides (AMPs) are emerging as the answer.
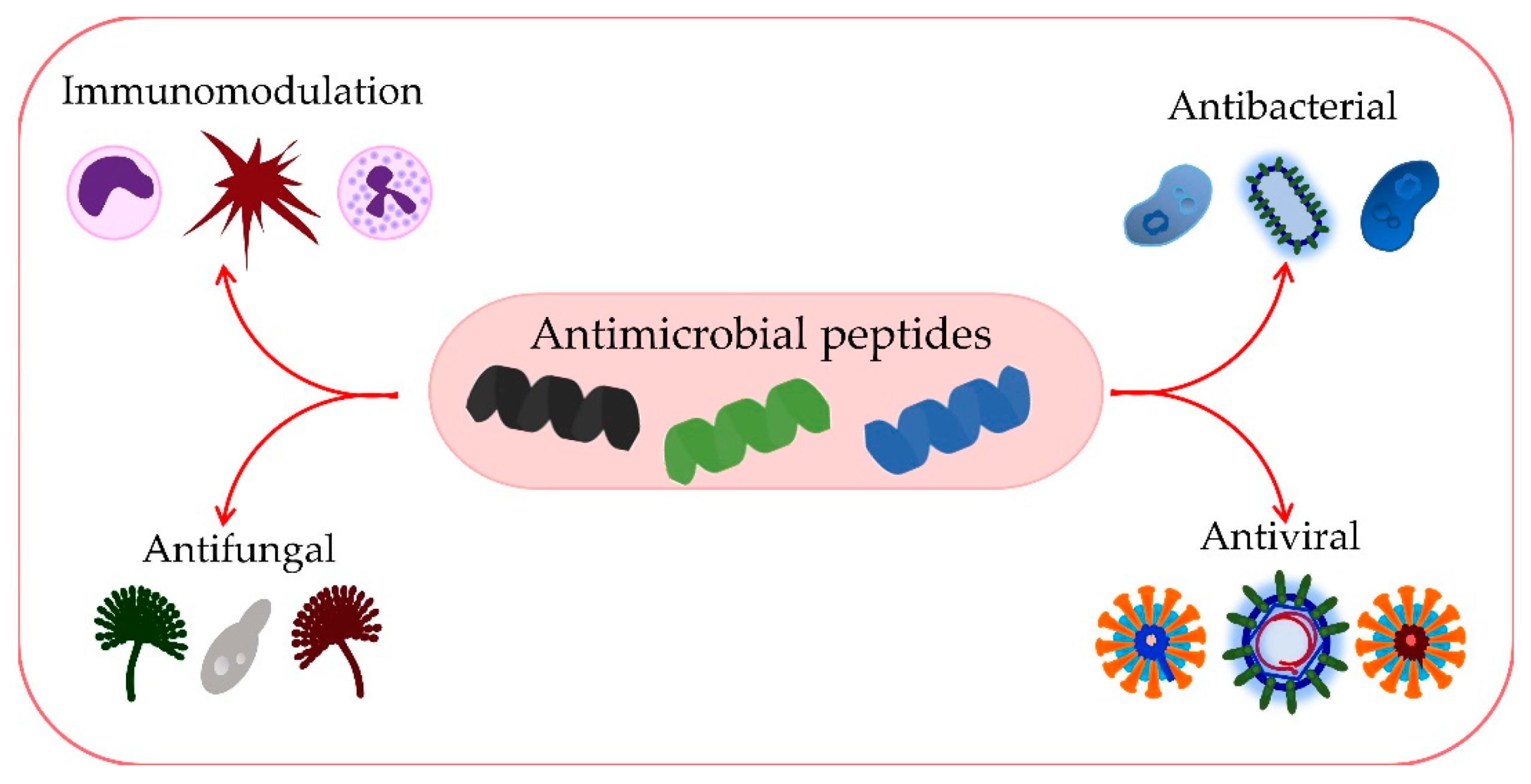
What Are Antimicrobial Peptides?
AMPs are small, naturally occurring peptides that form part of the innate immune systems of a wide range of organisms. When produced by bacteria themselves, they are classified as bacteriocins — essentially natural bacterial immune weapons that allow specific bacteria to inhibit competitors and maintain microbial stability.
This selectivity is what makes them so valuable in animal nutrition. Rather than resetting the gut environment indiscriminately, bacteriocins target specific threats while allowing beneficial flora to remain and flourish.
How ALMAT Works
ALMAT’s active bacteriocin is engineered for stability — a key differentiator. Most proteins are rapidly neutralised in the digestive tract before they reach their target. ALMAT’s unique molecular architecture allows it to remain active throughout both the farm environment and the full length of the gastrointestinal tract.
Its mechanism of action is highly specific: ALMAT binds to the secondary channel of bacterial RNA Polymerase, halting the initiation of RNA synthesis and causing rapid death of the target bacteria. Critically, this affects pathogens without disrupting the broader gut microbiome.
The result is what AND describes as mucosal immunity training — the animal’s own immune system is allowed to mature and strengthen, rather than being repeatedly overridden by broad-spectrum treatments.
Five Reasons to Use ALMAT
1. Antimicrobial activity — potent inhibition of Gram-negative pathogens including E. coli and Salmonella, with notable activity against Listeria monocytogenes.
2. Anti-biofilm effect — actively reduces biofilm formation from P. aeruginosa and Listeria, which are notoriously difficult to eliminate in farm environments.
3. Gut barrier protection — strengthens intestinal integrity by increasing tight junction expression, directly addressing leaky gut.
4. Immunomodulation — supports natural defences by modulating cytokine expression and reducing unproductive inflammation.
5. Performance promotion — improved feed efficiency, higher growth rates, and better litter quality through gut stability.

Practical Application
ALMAT is highly flexible in how it can be administered — drinking water, premix, or soluble feed — making it straightforward to integrate into existing nutritional programmes.
It is particularly recommended during high-stress periods: weaning, transport, and environmental changes, or whenever Gram-negative bacterial pressure is a concern. A minimum of 7 days is recommended for prevention, and 14 days for treatment.
Target species include piglets, poultry, and pre-ruminants.
One further practical advantage: ALMAT requires no withdrawal period when used according to regulations. This gives producers genuine flexibility to respond to market price peaks or slaughterhouse schedules without being locked into a treatment window.
Please refer to product documentation for specific usage and safety information. Contact your Agrihealth technical representative for further guidance.
